In the intricate web of human health, few connections are as fascinating and impactful as the gut-brain axis. This bidirectional communication network links our digestive system with our central nervous system, influencing everything from mood and cognition to immune function and chronic disease. I’ve tried to distill some studies and tie them into the foundational principles of osteopathic medicine, which emphasizes the body’s interconnectedness through its various systems. This post explores how understanding and nurturing this axis can enhance overall well-being, with a focus on osteopathic approaches that promote holistic healing.
The Anatomy of the Gut-Brain Axis: Building Blocks of Connection
At its core, the gut-brain axis relies on a sophisticated anatomical framework. The gastrointestinal tract, spanning about 8 meters in adults, originates from embryonic germ layers: the endoderm forms the epithelium and glands, the mesoderm contributes to connective tissues, smooth muscles, and blood vessels, while the ectoderm gives rise to the enteric nervous system (ENS) via neural crest cells. The gut divides into foregut, midgut, and hindgut segments, each with specific arterial supplies—the celiac artery for the foregut (including stomach and upper duodenum), superior mesenteric for the midgut (jejunum, ileum, and parts of the colon), and inferior mesenteric for the hindgut (descending colon and rectum). Proper midgut rotation during development prevents anomalies like malrotation, ensuring efficient nutrient absorption and waste elimination.
Central to this axis is the ENS, often called the “second brain,” embedded in the gut wall with over 100 million neurons—more than in the spinal cord. It comprises the myenteric plexus, which regulates peristalsis, and the submucosal plexus, which controls secretion and blood flow. The vagus nerve (cranial nerve X), the longest in the body, serves as the primary conduit, extending from the brainstem to the intestines, heart, and lungs. Its afferent fibers (gut-to-brain) vastly outnumber efferent ones (9:1 ratio), allowing sensory signals from the gut to influence brain function. Sympathetic innervation from thoracic segments T5-L2 further modulates gut activity, with specific overlaps like T5-7 for organs such as the liver and pancreas.
This anatomical setup isn’t isolated; it’s intertwined with the gut microbiome—a community of over 100 trillion microorganisms outnumbering human cells 3:1. These microbes, primarily Firmicutes and Bacteroidetes, reside in the intestinal lumen and interact with mucosal barriers, immune cells, and neural pathways. Disruptions here, such as reduced microbial diversity, can cascade through the axis, affecting distant systems.
To visualize this interplay, consider the following diagram illustrating key pathways:
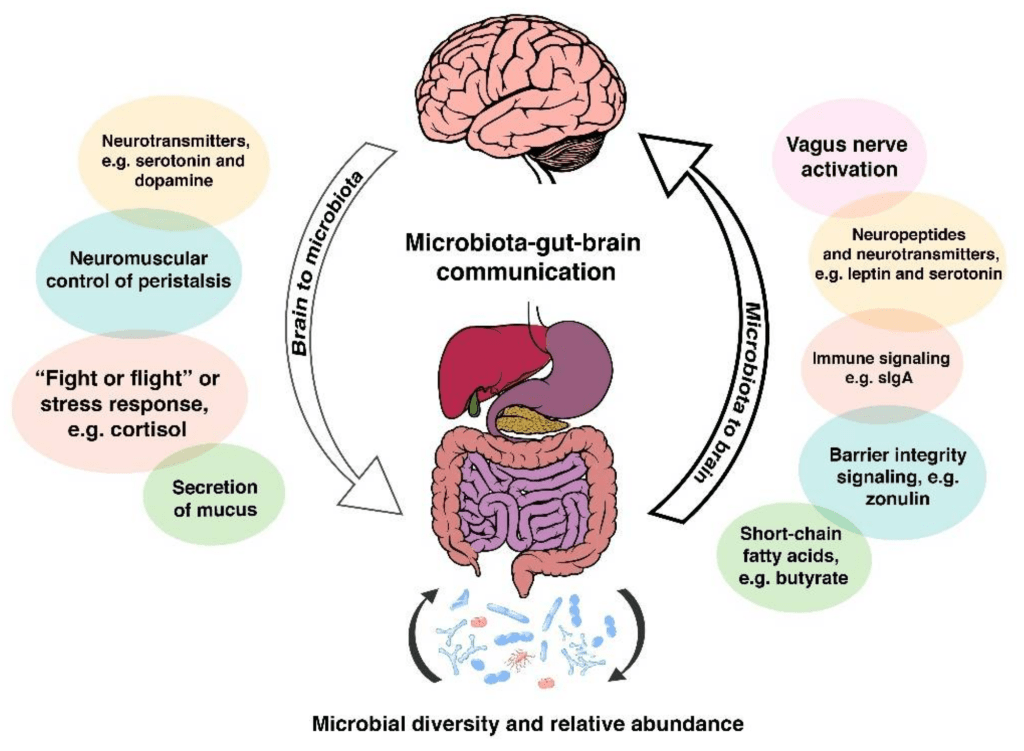
Physiology: The Dynamic Interplay of Signals
Physiologically, the gut-brain axis operates through neural, hormonal, immune, and microbial channels, creating a feedback loop that maintains homeostasis. The ENS produces over 30 neurotransmitters, including 95% of the body’s serotonin, which regulates mood, appetite, and gut motility. Microbial byproducts like short-chain fatty acids (SCFAs, e.g., butyrate) from fiber fermentation cross the blood-brain barrier, modulating inflammation and neuronal activity. For instance, SCFAs enhance barrier integrity by regulating proteins like zonulin, preventing “leaky gut” that could lead to systemic inflammation.
The vagus nerve facilitates rapid communication: gut microbes stimulate it, potentially boosting oxytocin release and influencing stress responses via the hypothalamic-pituitary-adrenal (HPA) axis. Stress hormones like cortisol, in turn, alter gut permeability and microbial composition, perpetuating a cycle. Immune signaling adds another layer; dysbiosis—imbalance in microbiota—promotes cytokine release, which can cross into the brain, exacerbating neuroinflammation in conditions like depression or Alzheimer’s disease. Hormonal pathways, including neuropeptides like leptin and serotonin, further link appetite control with emotional states.
In essence, a healthy axis promotes resilience: diverse microbiota aid digestion, immune regulation, and detoxification, while anatomical integrity ensures efficient signal transmission. Disruptions, often from diet, stress, or medications, can manifest as gastrointestinal disorders (e.g., IBS, IBD) or neurological issues (e.g., anxiety, cognitive decline), highlighting the body’s unified physiology.
Research Insights: Distilling the Evidence
Recent studies underscore the gut-brain axis’s role in health and disease. For example, research on dietary impacts shows that high-fiber, fermented, and calorie-restricted diets can alleviate mild anxiety and depression by modulating the microbiota. In inflammatory bowel disease (IBD), dysbiosis leads to loss of SCFA-producing bacteria, fueling mucosal inflammation and neuroimmune signals that disrupt mood via the axis. A 2025 review emphasizes how microbial metabolites, vagal tone, and the HPA axis form a loop perpetuating inflammation and altering mental health.
In neurodegenerative contexts, gut microbiota influence microglial activation in Alzheimer’s, with pro-inflammatory taxa increasing and beneficial ones decreasing, potentially serving as early biomarkers. Diet emerges as a key modulator; Western diets reduce diversity, linking to obesity and colorectal cancer, while interventions like probiotics restore balance. These findings, drawn from PubMed-indexed research up to 2026, reveal the axis as a therapeutic target, with microbial signatures predicting disease progression.
Integrating Osteopathic Medicine: A Hands-On Approach
Osteopathic medicine uniquely bridges anatomy, physiology, and the gut-brain axis by viewing the body as an interconnected unit. Osteopathic manipulative treatment (OMT) restores balance by addressing restrictions in blood flow, nerve supply, muscle tension, and organ mobility. For the gut, techniques target sympathetic tone at T5-L2, releasing ligament tensions and optimizing autonomic function.
Specific methods include indirect diaphragm release and rib raising to enhance lymphatic drainage and oxygen supply, reducing abdominal congestion. Mesenteric lifts improve venous return, bowel motility, and nutrient absorption, proving beneficial in conditions like IBS or post-traumatic stress disorder. Visceral osteopathy gently releases organ tensions, while sphincter treatments (e.g., for the ileocecal valve or pylorus) use clockwise motions to normalize function, aiding microbiome communication.
In IBD, OMT complements diet and fecal microbiota transplantation by modulating the axis, reducing neuroinflammation. Courses on the brain-gut-biome axis equip osteopaths to palpate microbiome-related dysfunctions, differentiating somatic, visceral, or microbial origins for precise interventions. This holistic rationale—rooted in osteopathy’s emphasis on self-healing—ties directly to anatomical pathways like the vagus nerve, promoting physiological harmony without over-reliance on pharmaceuticals.
Practical Applications for Everyday Health
To harness the gut-brain axis, start with diet: prioritize fiber-rich, diverse plant foods to boost microbial diversity and SCFA production. Probiotics and prebiotics can support balance, while stress management—through mindfulness or yoga—calms the HPA axis. For those with symptoms, consult an osteopathic physician for OMT, which can alleviate gut restrictions and enhance neural communication.
In summary, the gut-brain axis exemplifies the body’s unity, where anatomical structures enable physiological dialogues influenced by microbiota. Osteopathic medicine offers rational, hands-on tools to optimize this system, backed by emerging research.